TORONTO – RAMM Pharma Corp. (including its wholly owned subsidiaries, the “Company” or “RAMM”) (CSE: RAMM), a leader in plant-derived cannabinoid pharmaceutical products, is pleased to announce that the Uruguayan Ministry of Public Health, (Ministerio de Salúd Publica or “MSP”) has selected RAMM as a fulfilment and distribution partner for Uruguay’s COVID-19 vaccination program. Under the arrangement, RAMM will be responsible for the sterilization, packaging and distribution of syringes and other medical supplies for use in the country’s COVID-19 vaccination program. RAMM’s state-of-the-art GMP certified pharmaceutical facility was selected for the program upon satisfaction of the stringent requirements of the Ministry of Public Health.
“We are pleased to play an integral role in Uruguay’s COVID-19 vaccination effort. Our selection by the Ministry of Public Health is a testament to our longstanding relationship and recognition of the specialized capabilities of our team and GMP facility within the pharmaceutical community,” stated Jack Burnett, Chief Executive Officer.
RAMM’s wholly owned, state-of-the-art GMP certified formulation facility is ideally situated within close proximity to an airport and other distribution hubs. The facility totals approximately 36,600 square feet and features dedicated cannabis and medical product laboratories, along with sterilization and packaging capabilities and storage/distribution facilities.
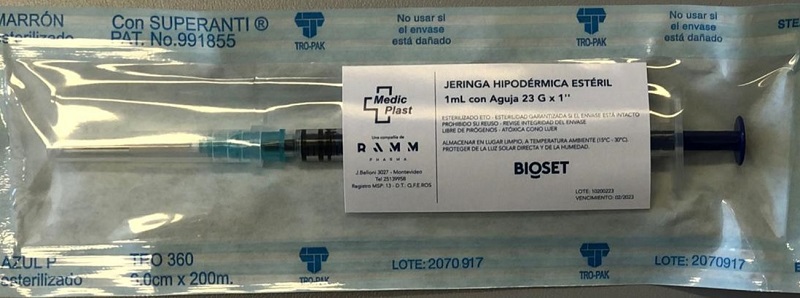
RAMM is an essential provider of medical and pharmaceutical products for the healthcare industry. Since it’s inception in 1988, RAMM’s wholly owned subsidiary Medic Plast SA has manufactured and supplied medical devices, antiseptic and sterile products and other medical supplies and continues to be a strategic supplier to the medical community in Uruguay. With a current portfolio of over 150 products registered with the Ministry of Health, Medic Plast is an established leader in the Uruguayan market for sales of recognized quality products under the BIOSET brand, including alcohol-based hand sanitizer and antiseptic soaps, sterile hospital devices and sterilization equipment, syringes and various other surgical and medical supplies.
About RAMM Pharma Corp.
Led by renowned cannabis industry experts and backed by successful pioneers in the cannabis sector, RAMM is a leader in the field of cannabinoid pharmacology and product formulation for cannabis-based pharmaceuticals and other cannabis-based products. Founded in 1988 in Montevideo, Uruguay, the Company is a well established pharmaceutical and medical product business that has developed medically registered and approved plant-derived cannabinoid pharmaceutical products. The Company currently has multiple approved and registered products that have been authorized for sale in Uruguay and compassionate use in several Latin American countries, as well as a pipeline of new products in various stages of approval and development produced in the Company’s state of the art Good Manufacturing Practice (GMP) certified cannabis formulation facility. Further to its industry leading activities in the cannabis sector, the Company operates a successful pharmaceutical, cosmetic and nutraceutical product development and medical services business which has been servicing the local market for 30 years. RAMM Pharma Corp. includes wholly owned subsidiaries Medic Plast SA, Yurelan SA, Glediser SA and Ramm Pharma Holdings Corp. Additional information about the Company is available at www.rammpharma.com.












